5% diclofenac ogwu maka eji anumanu
Njikọta
ML ọ bụla nwere diclofenac sodium 50mg
Njirimara:
Diclofenac sodium, sodium 2 - (2,6-Dichlorophophylyl) Phenocetate bụ otu n'ime ọgwụ mgbochi mgbochi mgbochi (nsaid), bụ nke acyl acetic acid dị na ya. Diclofenac sodium dị ka mgbochi mkpali, ihe ndị na - arịa antipmmac bụ n'ihi mbelata nke prostaglan synsation site na mgbochi site na mgbochi nke cycloid. Mmetụta ya bụ 2 ~ 25 ugboro 20 siri ike karịa indomotacin na 26 ~ ugboro 50 siri ike karịa acetylsaylicy acid. A mara ya na nrụpụta ọgwụ siri ike, mmeghachi omume dị nro, obere doses, na obere ọdịiche dị n'etiti.
Ọnụego ọgwụ ọgwụ na plasma protein bụ 99.7%. Ọkara ndụ bụ elekere 1 ruo 2. Dabere na dose akwadoro na nkeji ole na ole, enweghị nchịkọta. Imeju na imeju na - emeju na 60% nke metabolic ego nke metabolit ka a na-awụpụta site na akụrụ, na excretion nke ọgwụ mbụ ahụ erughị 1%. Ndị fọdụrụ n'ime ụdị nke metabolites, excreted na eriri afọ site na bile, ma kpochapụ site na anụ ahụ.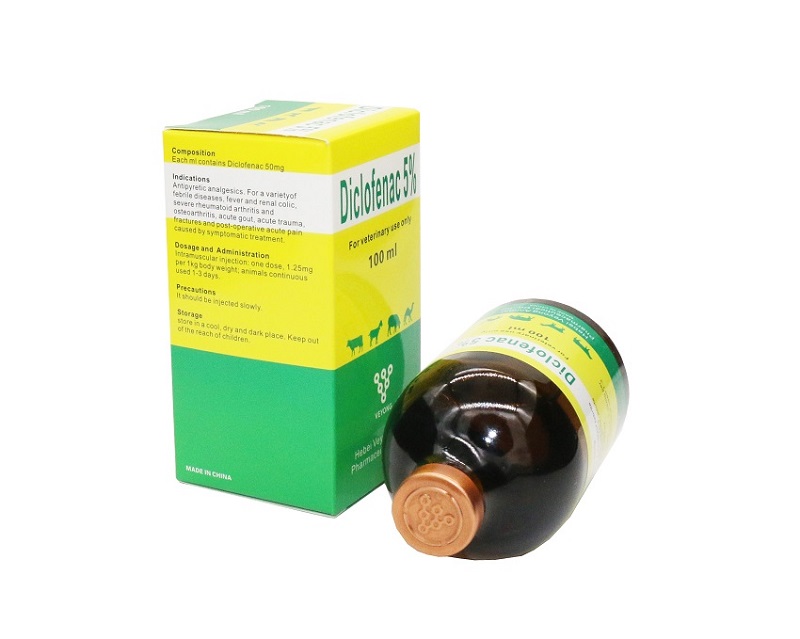
Ngosiputa
Antipyriatic. DICLOFOFOFOFENAC injection na mgbochi mkpali na analget, egosipụtara maka nchịkwa nke mgbu na mbufụt nke metụtara musclo-sọsọ na-enwe nsogbu dị ka; Ọrịa ogbu na nkwonkwo, osteochondritis, onye na-eme, ternsitis na myyositis, ọrịa oyi tutu ọ bụ mastitis na patitis.
A na-ejikwa ya maka njikwa fever metụtara ọrịa ma ọ bụ anụ ahụ anụ ahụ dị iche iche anụmanụ.
Usoro onunu ogwu na ochichi
Infomuscular ntụtụ:
otu dose, 1.25mg kwa ibu 1kg;
ụmụ anụmanụ na-aga n'ihu ụbọchị 1-3.
Mkpachapụ anya:Ọ ga-etinye ya nwayọ.
Storaji: Chekwaa na ebe dị mma, akọrọ ma gbaa ọchịchịrị. Hapụ ụmụaka aka.
Hedu Veyong Staycumcy Co., LTD, nke dị na Shijezhuan City, Hebei Provice, China, na-esote isi obodo Beijing. Ọ bụ ya buru ibu GMP na-emegwara banyere ọgwụgwọ ọgwụgwọ anụmanụ, na R & D, mmepụta na ire ahịa APA, nkwadebe maka faili na-eri nri. Dịka ụlọ ọrụ teknụzụ mpaghara, Veyong guzobere usoro R & D mepụtara ọhụrụ, ma bụrụ ndị ọkachamara na teknụzụ dị iche iche nke mba ahụ, enwere ndị ọkachamara 65. Veyog nwere ntọala abụọ na-emepụta ihe: Shinazhung Bag na ọdụ nke Shinmectin na-ekpuchi, exines, ntụ ntụ, bolucides na disinfter, ects. Veyong na-enye APIS, ihe karịrị 100 nke onwe ya na nkwadebe ya, yana ọrụ OEM & ODM.
Veyong na-etinye oke mkpa maka njikwa EHS (gburugburu ebe obibi, ahụike & nchekwa) sistemụ, ma nweta asambodo ISO14001 na Ohsas18001 asambodo. Edepụtala Veyong edepụtara na ụlọ ọrụ mmepụta ụlọ ọrụ na HEBIEI na - enwe ike ịgba mbọ hụ na ngwaahịa ngwaahịa na - aga n'ihu.

Asambodo Asambodo zuru ezu, Asambodo ISO9001, Asambodo Asambodo GMP, Atiopia Gmp Gmp, Atiopia GMP, Ahịhịa GMP, ma gafere nyocha US FDA. Veyong nwere ndị otu ọkachamara na ndebanye aha, ọrụ na ọrụ aka ọrụ, ụlọ ọrụ anyị enwetala ntụkwasị obi na nkwado ngwaahịa dị mma, ụgwọ ahịa dị elu na ọrụ ọpụpụ na nke sayensị. Veyong emeela nkwado ogologo oge na ụlọ ọrụ ọgwụ ụmụ anụmanụ ama ama na ngwaahịa ndị na-ebuga na ngwaahịa ndị a na-ebugharị na Europe, Middle Africa, wdg mba 60 na mpaghara.




.png)
.png)
.png)
.png)